Artículo de Investigación 
Maternal risk factors in full-term newborns with early onset sepsis related with Interleukin-6 in umbilical blood
Fabricio González-Andrade1, Andrea Castillo Rojas2, Gabriela Aguinaga Romero3
1 Fabricio González-Andrade, MD, PhD. Translational Medicine Unit, Faculty of Medical Sciences at Central University of Ecuador. Quito, Ecuador. ORCID: 0000-0002-2091-9095. Correo e.: fabriciogonzaleza@gmail.com. Corresponding author.
2 Andrea Castillo Rojas, MD. Translational Medicine Unit, Faculty of Medical Sciences at Central University of Ecuador. Quito, Ecuador. ORCID: 0000-0002-4878-4700. Correo e.: enana_agcr@hotmail.com
3 Gabriela Aguinaga Romero, MD. Translational Medicine Unit, Faculty of Medical Sciences at Central University of Ecuador. Quito, Ecuador. ORCID: 0000-0002-6274-2195. Correo e.: gabrielaaguinaga@yahoo.com
Archivos de Medicina (Manizales) Volumen 19 N° 2, Julio-Diciembre 2019, ISSN versión impresa 1657-320X, ISSN versión en línea 2339-3874, González Andrade F., Castillo Rojas A., Aguinaga Romero G.
Recibido para publicación: 07-04-2019 - Versión corregida: 18-07-2019 - Aprobado para publicación: 24-07-2019
González-Andrade F, Castillo-Rojas A, Aguinaga-Romero G. Maternal risk factors in full-term newborns with early onset sepsis related with Interleukin-6 in umbilical blood. Arch Med (Manizales) 2019; 19(2):320-30. DOI: https://doi.org/10.30554/archmed.19.2.3298.2019
Summary
Objective: to determine the relationship and association of early sepsis with interleukin-6 measured in umbilical blood, as well as gestational age and birth weight, prenatal history of the mother pathologies during pregnancy, in newborns at the Neonatology Service of Carlos Andrade Marín Hospital (Quito). Material and methods: epidemiological, observational, analytical, cross-sectional. 200 newborns were analyzed. IL-6 was determined by Elisa. Results: newborn mean gestational age 38.4±2 weeks, mean weight 2871±532.4 grams, cutoff point interleukin 6 ≤14pg/uL; male sex was 43.9%, female sex 56.1%; preeclampsia of the mother 22.4%, infection of the urinary tract during pregnancy 20.4%; vaginitis/vaginosis 19.4%; IL6 values ≤ 14 pg/uL in 63.3% of patients; IL6 values of >14 pg/uL 36.7%; relationship with sepsis in all, p<0.05; relationship of prenatal pre-maternity of the mother and neonatal sepsis, p<0.05; relationship between vaginitis/vaginosis of the mother with neonatal sepsis; p<0.05; OR: 38.88 IC95% (4.912-307.728); association between sepsis and risk factors, IL6, gestational age in weeks, weight, prenatal preeclampsia and urinary tract infection, vaginitis/vaginosis, p<0.05. Conclusion: it is concluded that there is a correlation of risk factors, such as gestational age in weeks, weight, prenatal preeclampsia, UTI prior to birth, vaginitis/vaginosis, and values of interleukin 6 ≤ 14 pg/uL. The mean gestational age was 38.4 weeks, and average weight 2871 grams in the newborns analyzed.
Keywords: risk factors, biomarkers, interleukin-6, pre-eclampsia, newborn, neonatal sepsis.
Factores de riesgo maternos en recién nacidos a término con sepsis de aparición temprana relacionada con interleucina-6 en sangre umbilical
Resumen
Objetivo: determinar la relación de sepsis temprana con interleucina 6 medida en sangre umbilical, así como la edad gestacional y el peso al nacer, antecedentes prenatales de patologías maternas durante el embarazo, en recién nacidos en el Servicio de Neonatología del Hospital Carlos Andrade Marín (Quito). Materiales y métodos: estudio epidemiológico, observacional, analítico, transversal. Se analizaron 200 recién nacidos. La IL-6 fue determinada por Elisa. Resultados: edad gestacional promedio del recién nacido 38.4±2 semanas, peso promedio 2871±532.4 gramos, punto de corte interleucina 6 ≤14pg/uL; el sexo masculino fue del 43,9%, el sexo femenino del 56,1%; preeclampsia de la madre 22.4%, infección del tracto urinario durante el embarazo 20.4%; vaginitis/vaginosis 19,4%; Valores promedio de IL6 ≤14 pg/uL en el 63,3% de los pacientes; valores de IL6 de> 14 pg/uL en 36.7% de pacientes; relación con sepsis en total, p<0.05; relación de la pre-maternidad prenatal de la madre y sepsis neonatal con p <0.05; relación entre vaginitis/vaginosis de la madre con sepsis neonatal con p<0.05; OR (verosimilitud): 38,88 IC95% (4.912-307.728); asociación entre sepsis y factores de riesgo, IL6, edad gestacional en semanas, peso, preeclampsia prenatal e infección del tracto urinario, vaginitis/vaginosis, p <0,05. Conclusión: se concluye que existe correlación de factores de riesgo como la edad gestacional en semanas, el peso, la preeclampsia prenatal, la infección de vías urinarias antes del nacimiento, la vaginitis/vaginosis y los valores de interleucina 6 ≤1 4 pg/uL. La edad gestacional media fue de 38,4 semanas y el peso promedio de 2871 gramos en los recién nacidos analizados.
Palabras Clave: factores de riesgo, biomarcadores, interleucina-6, pre-eclampsia, recién nacido, sepsis neonatal.
Introduction
Neonatal sepsis is a systemic infection occurring in infants at ≤28 days of life, and it is an important cause of newborn morbidity and mortality. The signs and symptoms are nonspecific and make diagnosis difficult; routine tests are not completely effective, are nonspecific, and cause the inappropriate use of antibiotics [1]. Sepsis is the result of an infection associated with a systemic inflammatory response and with production and release of a wide range of inflammatory mediators. It is necessary to analyze cytokines [2] which are potent inflammatory mediators. Serum levels also increase during infections, therefore changes can occur from other inflammatory effector molecules. Group B streptococcus (GBS) is the most common etiologic agent, while Escherichia coli is the most common cause of mortality [3].
The diagnosis of neonatal sepsis is based on a combination of clinical presentation; the use of nonspecific markers, including C-reactive protein (CRP) and procalcitonin (PCT), and blood cultures [4]. Cytokines, including interleukin 6 (IL-6), interleukin 8 (IL-8), gamma interferon (IFN-γ), and tumor necrosis factor alpha (TNF-α), and cell surface antigens, including soluble intercellular adhesion molecule (sICAM) and CD64, are also used as nonspecific screening measures for neonatal sepsis [6].
Neonatal sepsis is a clinical situation that requires rapid action and the establishment of adequate antimicrobial therapy. The initial symptoms are usually nonspecific; respiratory manifestations are the first to appear. Neonatal sepsis has an incidence of 1-10 cases per 1,000 live births and a high mortality rate. IL-6 has a sensitivity of 87-100%, a negative predictive value between 93 and 100% [61]. It is effective for the diagnosis of late neonatal sepsis in preterm infants. At the beginning of the infection it has a high sensitivity (89%) and a negative predictive value of 91% [7].
Cytokines are proteins secreted by the cells of innate and adaptive immunity that mediate cellular functions. Cytokines have pleiotropic and redundant action, and are secreted freely and self-limited. They influence the synthesis and actions of other cytokines, may have local and/or systemic action, and initiate their actions by joining specific membrane receptors in the target cells. Cytokines are regulated by external signals, and cause changes in the gene expression of target cells and have inhibitory mechanisms of feed-back [8].
Interleukin 6 (IL-6) is a cytokine that is synthesized by mononuclear phagocytes, vascular endothelial cells, fibroblasts, and other cytokines such as IL1 and TNF. The functional form and IL6 is a homodimer, and each subunit forms a globular domain with four alpha helices. The IL6 receptor is formed by a cytokine binding protein and a subunit for signal transduction, and both belong to the cytokine receptor family of type I [9]. IL-6 has several actions. In innate immunity, it stimulates the synthesis of proteins of the acute phase by hepatocytes, and also stimulates the production of neutrophils from progenitors of the bone marrow in synergy with colony stimulating factors. In adaptive immunity, it stimulates the growth of lymphocytes B, producers of antibodies, lymphocytes T, and induces fever. In addition, IL-6 acts as an autocrine growth factor in neoplasic cells. It also stimulates the synthesis of other proinflammatory cytokines, especially IL-17 [10].
According to some studies, IL-6 could play a key role in the inflammatory response to microbial pathogens. In the same way, a high IL-6 level was associated with increased severe sepsis mortality and risk, and IL-6 has also been investigated among other infection markers to improve the diagnostics of suspected neonatal infection. Furthermore, IL-6 signal pathway was essential to systemic inflammation in mice studies. For this reason, it could be possible to be related to a gene for sepsis [11]. A systemic fetal cytokine response, defined as a plasma interleukin-6 (IL-6) value above 11 pg/m is considered a major independent risk factor, such as fetal inflammatory response syndrome (FIRS), a diagnostic marker of early-onset neonatal sepsis (EONS) and a risk predictor of white matter injury (WMI) [12].
Persistently elevated levels of IL-6 in plasma below peak values are considered predictors of poor prognosis in patients with septic shock [13]. Increased levels of plasma IL-6 in patients with sepsis correlate with the severity of septic shock, and an increase in serum levels of IL-6 during sepsis is associated with an increased incidence of shock and worse prognosis. IL-6 seems to be a better parameter to assess the severity of sepsis, occurring as early as two to four hours after the onset of the inflammatory response [14].
The aim of this research is to determine the relationship and association of early sepsis with interleukin-6, measured in umbilical blood, as well as gestational age and birth weight, and prenatal history of the mother in full-term newborns with and without risk factors.
Material and methods
Research design. Epidemiological, observational, analytical, cross-sectional design.
Sampling: it analyzed two cohorts of patients, born at the Neonatology Service of the Carlos Andrade Marín Hospital (HCAM), located in the Metropolitan District of Quito, Ecuador; through 2016 and 2017. Samples correspond to full-term newborns with risk factors for early sepsis (n=100), and full-term newborns without risk factors (n=100). It used finite universe sampling calculations, however, the number of patients obtained was less than 100, for that reason, it selected randomly two cohorts of 100 patients to reach a discrimination power of 80%.
Inclusion criteria: patients with risk factors, patients whose mothers have had urinary tract infections (UTIs) without treatment, or other related infections. Exclusion criteria: patients older than 37 weeks without risk factors, patients whose mothers have not presented any infection, or received treatment. Criteria for elimination: patients who have died post-partum by any clinical condition.
Specific methods: an information collection sheet was used to gather all information required. IL-6 was determined by conventional electrochemiluminescence immunoassay (ECLIA) method, at the Central Laboratory in the hospital.
Ethical criteria: approval was obtained from the Ethics Committee in Human Research of the hospital, and by the Institutional Review Board (IRB) of the Central University of Ecuador. Information was collected from a secondary source (medical records), therefore, no informed consent was required. In terms of confidentiality, the protection of the identity of the persons participating in the investigation was ensured, the study has no potential risks, and it provides benefits in future applications.
Data analysis: data collection, analysis and evaluation were collected in an Excel database to be analyzed with the IBM SPSS© software version 22, licensed. It used descriptive statistics, measures of central tendency, and measures of variability (spread). Measures of central tendency include the mean, median, and mode, while measures of variability include the standard deviation, variance, the minimum and maximum variables, and the kurtosis and skewness. The relationship model of the observed variables with neonatal sepsis was analyzed using binary logistic regression.
Results
Distribution of newborn sex, risk factors, sepsis and IL-6. 200 children were analized, including males (43.9%) and females (56.1%). In relation to the mother’s medical history that constitutes risk factors for sepsis for the child, 22 showed prenatal preeclampsia (22.4%), and 20 showed UTI (20.4%). Likewise, prenatal vagintis and vaginosis were found in 19 mothers (19.4%). 43 children (43.9%) developed sepsis. In relation to IL-6, 62 children had ≤ 14pg/uL (63.3%) while 36 children showed IL-6 >14 pg/uL (36.7%). In the relationship with sepsis, statistical significance was found p<0.05 with all the variables, except with newborn sex. p>0.05. See Table 1.
Table 1. Distribution of newborn sex, risk factors, sepsis and interleukin-6.
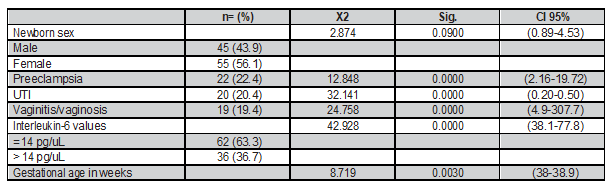
Source: Neonatology Service. Hospital Carlos Andrade Marín (Quito, Ecuador).
Distribution of the most important findings related to weight, gestational age in weeks, and IL6. An average weight of 2870.7±532.4 grams was found in the neonates. The minimum weight was 1015 g and the maximum of 4180 g. The P25 was 2611.3 g. and the P50 of 2950 g. The P63 is 3106.7 g and corresponds to the cutoff point for IL-6, of ≤ 14 pg/ul, P75 is 3207.5 g. The average gestational age is 38.4±2 weeks; the lowest value was 31 weeks and the maximum was 40 weeks, with a range of 19 weeks, the P25 is 37.2 weeks. P50 38.6 weeks. P63 corresponds to 39.2 weeks, and is related to the cut of interleukin 6, of ≤14 pg/ul. P75 is 39.7 weeks; the mean of interleukin 6 is 58±99.1 pg/ul, the minimum value is 0 and the maximum value is 414 pg/ul, the P25 is 0.1 pg/ul, the P50 is 0.4 pg/ul. P75 is 101.5 pg/ul. See Table 2.
Table 2. Distribution of the most important findings related to weight, gestational age in weeks and interleukin-6.
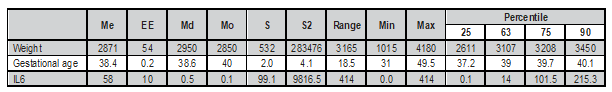
Me=average; EE=standard error; Md=median; S=standard deviation; S2=variance.
Source: Neonatology Service. Hospital Carlos Andrade Marín (Quito, Ecuador).
Distribution of the relationship between sepsis with prenatal preeclampsia, vaginitis, prenatal vaginosis, and newborn sex. In the study of prenatal infections and neonatal sepsis, it can be seen that of 43 cases of sepsis, 17 mothers of neonates who developed sepsis (39.5%) presented prenatal preeclampsia, and 26 (60.5%), despite newborn sepsis, did not present preeclampsia before birth. Of the 55 neonates who did not have sepsis, five of their mothers (9.1%) had prenatal preeclampsia and 50 mothers of neonates (90.9%) did not present preeclampsia.
The relationship between vaginitis or prenatal vaginosis of the mother with neonatal sepsis. Of 43 children with sepsis, 18 patients’ mothers had vaginitis or prenatal vaginosis (41.9%) and 25 patients presented sepsis (58.1%). 55 neonates did not present sepsis, despite the fact that one mother had a history of vaginitis or vaginosis (1.8%), and 54 neonates did not have sepsis (98.2%) and neither did the mother have a history of vaginitis or prenatal vaginosis; statistical significance was found (p=0.000). When a mother has vaginitis or vaginosis there is 39 times more risk of developing neonatal sepsis. OR: 38.88 IC95% (4.912-307.788). Concerning the link between sex and sepsis, of 43 patients with sepsis, 23 children (53.5%) corresponded to male sex and 20 to female sex (46.5%); 55 children did not develop sepsis including 20 boys (36.4%) and 35 girls (63.6%). No statistical significance was found p>0.05 (p=0.090). In the case of the relationship between UTI with sepsis, it was not possible to establish the contingency table, because a quadrant generated zero (0). However, it observed that of 43 children with sepsis, 20 mothers had a UTI (46.5%), and 23 children had no previous history of this pathology in their mothers (53.5%). In the case of IL-6 (dichotomized) and sepsis, a zero was also presented in one of the quadrants, but in the same way it was observed that of 43 children with sepsis, seven had interleukin ≤ 14 pg/ul (13.3%) and 36 children presented interleukin >14 pg/ul; while 55 children with interleukin ≤ 14 pg/ul did not have sepsis. See table 3.
Table 3. Distribution of the relationship between sepsis with prenatal preeclampsia vaginitis, vaginosis and newborn sex.
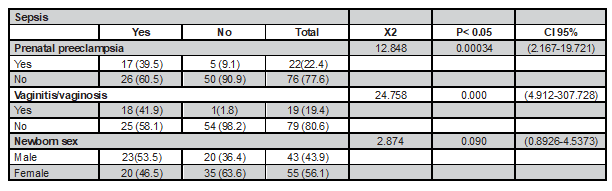
Source: Neonatology Service. Hospital Carlos Andrade Marín (Quito).
Relationship model of the variables with neonatal sepsis. Using binary logistic regression, statistical significance was found (p <0.05) in the connection between sepsis (endogenous variable) and the following exogenous variables (risk factors): IL-6 (p=0.0000), gestational age (weeks) (p=0.0030), weight (p=0.0000), prenatal preeclampsia (p=0.0000), prenatal UTI (p=0.0000), vaginitis/vaginosis (p=0.0000) and with sex. No statistical significance was found (p>0.05). Therefore, it is concluded that there is a relationship between sepsis and the exogenous variables, considered a risk factor for sepsis in neonates with statistical significance of p<0.05. See table 4.
Table 4. Relationship model of the variables with neonatal sepsis using binary logistic regression.
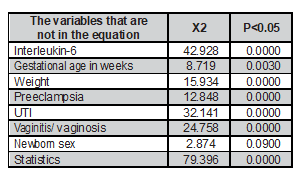
Source: Neonatology Service. Hospital Carlos Andrade Marín (Quito).
Discussion
Distribution of newborn sex, risk factors, sepsis, and interleukin 6.
A significant difference was found between sepsis and risk factors, such as pre-eclampsia in the prenatal period, infection of the urinary tract of the mother (UTI), vaginitis and/or prenatal vaginosis present in the mothers, and the quantification of IL-6 in umbilical cord blood less than 14 pg/uL. In addition, in this study there was a predominance of female sex, which contrasts with other studies where the predominance is male. This prevalence can be attributed to hormonal and genetic factors, and biological inflammatory markers related to the female sex, where they play a protective role. Another important factor is preeclampsia, which is attributed to oxidative stress and antioxidant status in neonates born to pre-eclamptic mothers. Another is sepsis due to infections of the urinary tract of the mother, in particular, bacteria that can invade and spread in the urinary system through the ascending lymphatic and hematogenous routes. All these factors contribute to low birth weight and/or a premature newborn, with strong evidence of intrauterine growth retardation.
On the other hand, sepsis, secondary to prenatal vaginosis, is attributed to vertical transmission, caused by germs located in the maternal genital canal that contaminate the fetus ascending up the birth canal to reach the amniotic fluid or by direct contact of the fetus with contaminated secretions also passing through the birth canal. It is well known that sepsis contributes to morbidity and mortality in the neonatal period, so that diagnosis and early treatment improve its prognosis and outcome. The usefulness of IL-6 in the diagnosis of neonatal, early, and late sepsis is based on the concentrations of different cytokines in cord blood and in peripheral blood. Therefore, it is attributed to cytokines that act non-enzymatically as intercellular signals that modulate cellular function in the inflammatory response, regulating the growth, motility, and differentiation of leukocytes and non-leukocyte cells. Its functions are performed by the autocrine effect, that is, on the cell that produces it; the paracrine effect on the microenvironment of the cell, or the endocrine effect with distal action. In summary, the most important factors that explain the in vivo effects of IL-6 are the magnitude of its production, its half-life, body distribution, and the presence of natural inhibitors.
In addition, several authors [14] report that septicemia is one of the main causes of mortality and morbidity in newborns, and establish that the increase in the level of IL-6 can occur before the C reactive protein (CPR) and can be used as a good index in the diagnosis of early sepsis compared to PC; therefore at the level of IL-6 is attributed its best determination of early sepsis compared to CPR6. Other study [16] says that the reference value of IL-8 was <14.1 pg/mL for healthy term infants administered vaginally and by cesarean section; for IL-6 the reference value is <10.2 pg/mL. Early-onset sepsis remains a common and serious problem for neonates, especially premature infants. In another study, on the clinical validity of IL-6 determinations in preterm infants, the authors conclude that the concentration values measured in real days can be considered an early marker of sepsis. Similarly [17], after studying the epidemiological characteristics of perinatal risk factors and neonatal outcomes, maternal age, pathologies of pregnancy and childbirth, as well as neonatal outcomes explain the risks of pathological conditions and the results of newborns are also related to the increase in IL-6 values.
One author [18] evaluated the diagnostic tests of inflammatory markers in maternal serum or umbilical cord blood for the diagnosis of onset of early neonatal sepsis in relation to diagnostic yield of procalcitonin (PCT), C-reactive protein (CRP), IL-6, and counting of leukocytes in umbilical cord blood or maternal serum for the diagnosis of early neonatal sepsis, obtaining similar conclusions. When they [19] studied groups of infection by Gram-negative, Gram-positive, and by fungi, they described the optimal cut-off levels of IL-6 between the groups of Gram-negative and Gram-positive infections were 202 and 57 pg/mL respectively. The fungal infection group had higher CPR levels than the infection groups with Gram bacteria, and concluded that Gram-negative organisms resulted in 10-fold higher IL-6 production than the other groups of bacteria.
The predictive value of PCT in umbilical cord blood and IL-6 in the diagnosis of early sepsis in premature infants has also been studied, and a negative correlation was found in the diagnosis of early sepsis with the presence of negative or positive clinical culture. The combination of PCT and IL-6 showed a sensitivity of 77% and specificity of 91.7%, which demonstrates its usefulness in the prediction of early sepsis. It also evaluated IL-6, IL-8, and IL-10 as possible markers of early diagnosis of neonatal infection and its relationship with the mortality and prognosis rate, and observed statistically significant differences between the control and case groups for the level of the three interleukins studied. They suggested a cut-off value of 10.85 pg/mL of IL-6, to discriminate between cases and controls, and a value of 78.2 pg/mL to predict mortality, and for IL-8 a cut-off value of 60 μg/mL was the value to differentiate definite versus indefinite infection. They attribute a greater potential to predict infection and possible mortality to IL-6, while IL-8 was valuable for the diagnosis of definitive infection.
On the other hand, concentrations of IL-6 have also been determined [20] during the microbial invasion of the amniotic cavity (MIAC) and amniotic fluid after delivery, and they evaluated the presence of acute histological chorioamnionitis (ACH) in the placenta. They also observed a greater intra-amniotic inflammatory response, which was mediated by IL-6 concentrations. It concluded that both MIAC and ACH affect the intensity of the intraamniotic inflammatory response and the incidence of early sepsis following subsequent delivery, to premature rupture of membranes before preterm labor between 34 and 37 weeks of pregnancy. They report that the fetal inflammatory response syndrome (FIRS) was defined with an IL-6 in umbilical cord blood greater than 11 pg/mL. The main parameter evaluated was a neonatal adverse result defined as in-hospital mortality and/or the presence of any of five pre-specified comorbidities, such as bronchopulmonary dysplasia, periventricular leukomalacia, intraventricular hemorrhage and early onset or onset of late sepsis [21]. All of them presented FIRS with mean values of IL-6 of 51.8 pg/mL.
Distribution of the findings related to weight, gestational age in weeks and interleukin 6
The present study found mean gestational age of 38.4±2 weeks, weight of 2871±532.4 g, corresponding to IL-6 of ≤14 pg/uL. in children with a low birth weight of 1015 g and a gestational age of 31 weeks, who are related to prenatal preeclampsia, prenatal UTI, vaginitis/vaginosis, IL6 reaches a value of up to 414 pg/uL, with the P90 percentile up to 215 pg/uL, which is attributed to the presence of early neonatal sepsis.
Several authors [22-24] state that the death rate due to sepsis is high and the risk of sepsis increases in prematurity in proportion to the decrease in birth weight; evaluation of levels of IL-6 and CPR for the diagnosis of early sepsis may explain the difference between septic and non-septic patients. Neonatal sepsis, characterized by systemic signs of infection in the first month of life, remains an important clinical syndrome, with high mortality and morbidity rates; the use of IL-6 and CRP in combination have been shown to be useful in the early diagnosis of sepsis in newborns. It was observed [25, 26] that umbilical cord IL-6 above the P75 percentile was associated with an increase in respiratory distress syndrome and chronic lung disease, but not with necrotizing enterocolitis, intraventricular hemorrhage or neonatal sepsis [28]. This association may change after adjustment for weeks of gestation at delivery and the treatment group. In our study, IL-6 P75 percentile, was of 101.5 pg/uL, and is associated with an increase in respiratory distress syndrome. Similarly, other studies demonstrate greater diagnostic accuracy in differentiating sepsis from non-sepsis when measuring IL-6 in combination with CRP.
About the distribution of the relationship between sepsis with prenatal preeclampsia, vaginitis/vaginosis and newborn sex
In the present study, a connection was found between prenatal preeclampsia and neonatal sepsis, as well as between vaginitis and vaginosis with neonatal sepsis. No connection was found between sex of the newborn and sepsis. There was a link discovered between UTI with sepsis, and the values of IL-6 with sepsis, with values lower of 14 pg/uL. Preeclampsia is a risk factor that is attributed to oxidative stress and antioxidant status in neonates born to pre-eclamptic mothers; sepsis due to UTI is due to the invasion and dissemination of bacteria in the urinary system through the described routes, which cause a child of low birth weight and premature newborn. Sepsis secondary to prenatal vaginosis is attributed to vertical transmission. Several authors report that all markers of oxidative stress, including IL-6, were higher in the preeclampsia group compared to controls [27].
The relationship model of the variables with neonatal sepsis
In the present study, binary logistic regression showed an association of sepsis with IL-6 and other risk factors such as gestational age, weight, and prenatal preeclampsia, infections of the urinary tract before birth, vaginitis, and vaginosis. Therefore, it concluded that IL-6 is a predictor of early sepsis associated with prenatal risk factors, previously described.
Prematurity is attributed as the main risk factor for sepsis, since it is associated with immaturity of the immune system. This is explained by the low levels of immunoglobulin related to the decrease of transplacental transfer of maternal IgG; the decrease in the function of neutrophils and monocytes that manifests itself with the alteration of the function of chemotaxis, opsonization and bacterial phagocytosis; and by a decreased function of the barrier of the skin and mucous membranes. Decreased Sepsis causes high mortality in newborns with low and very low birth weight.
Several authors [28] did report that the lipopolysaccharide binding protein (LBP), IL-6, and CRP were significantly higher in children with severe sepsis compared with those with less severe sepsis; the mean values of LBP, IL6, and CRP were significantly higher in children with bacteremia compared to those without bacteremia. Therefore, they conclude that elevated levels of LBP, IL-6, and CRP were associated with a more severe level of infection in children. It seems that these biomarkers are good markers to detect patients with bacteremia.
The IL-6 values of the umbilical cord and funisitis have also been evaluated, as an independent predictor of early-onset neonatal sepsis (EONS), in the presence of premature rupture of membranes. Funisitis was defined according to the classification of Salafia; the data were adjusted for gestational age at the time of delivery and prenatal administration of corticosteroids and antibiotics. IL-6 from the umbilical cord was found to be the only independent predictor of early-onset neonatal sepsis in premature rupture of preterm membranes, which was not observed in funisitis.
Currently, there is no adequate laboratory test, single or in combination, which has sufficient sensitivity and specificity to allow physicians to safely rule out early-onset sepsis.
All sick newborns with clinical signs suggestive of sepsis should be treated empirically with antibiotics, once the respective cultures have been taken. Treatment of term infants with a good outcome depends on the number of risk factors that are controlled, including maternal colonization of GBS. Maternal intrapartum antibiotic prophylaxis for GBS must be individualized. Careful evaluation and observation of these newborns with risk factors is a fundamental component of adequate care during the first 24 hours of life.
Conclusion. It is concluded that there is a relationship of risk factors, such as gestational age in weeks, weight, prenatal preeclampsia and UTI, vaginitis, vaginosis, and values of interleukin 6 of ≤ 14 pg/uL. The mean gestational age was 38.4 weeks, and average weight 2871 grams in the newborns analyzed.
Contribution of the authors
The research protocol and its design, data collection, statistical analysis, evaluation and interpretation of the data, critical analysis, discussion, writing and approval of the final manuscript were carried out by all authors who contributed in the same way in the whole process. The corresponding author represents the group of authors.
Availability of data and materials
The data supporting this manuscript are available upon request to the corresponding author.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committe and with the 1964 Helsinki Declaration and is later amendments or comparable ethical standars. Informed consent was obtained from all individual participants included in the study.
Consent for publication
The institutions cited in this document gave their consent to use this information.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest: no financial or nonfinancial benefits have been received or will be received from any party related directly or indirectly to the subject of this paper.
Funding source: the authors declare that the financial resources for the elaboration of the present investigation do not come from any fund, but from their self-management.
Cited literature
1. Hornik CP, Fort P, Clark RH, Watt K, Benjamin DK Jr, Smith PB, et al. Early and late onset sepsis in very-low-birth-weight infants from a large group of neonatal intensive care units. Early Hum Dev
2012; 88(Suppl 2):S69-74. DOI: 10.1016/S0378-
3782(12)70019-1
2. Simonsen KA, Anderson-Berry AL, Delair SF, Davies HD. Early-onset neonatal sepsis. Clin Microbiol Rev 2014; 27(1):21-47. DOI: 10.1128/ CMR.00031-13
3. Bedford Russell AR, Kumar R. Early onset neo- natal sepsis: diagnostic dilemmas and practi- cal management. Arch Dis Child Fetal Neonatal Ed. 2015; 100(4): F350-4. DOI: 10.1136/archdis- child-2014-306193
4. Fan Y, Yu JL. Umbilical blood biomarkers for predicting early-onset neonatal sepsis. World J Pediatr. 2012; 8(2):101-8. DOI: 10.1007/s12519-
012-0347-3
5. Chiesa C, Pacifico L, Natale F, Hofer N, Osborn JF, Resch B. Fetal and early neonatal interleu- kin-6 response. Cytokine 2015; 76(1): 1-12. DOI:
10.1016/j.cyto.2015.03.015
6. Su H, Chang SS, Han CM, Wu KY, Li MC, Huang CY, et al. Inflammatory markers in cord blood or maternal serum for early detection of neonatal sepsis-a systemic review and meta-analysis. J Perinatol 2014; 34(4):268-74. DOI: 10.1038/ jp.2013.186
7. Sugitharini V, Prema A, Berla Thangam E. Inflam- matory mediators of systemic inflammation in neonatal sepsis. Inflamm Res 2013; 62(12):1025-
34. DOI: 10.1007/s00011-013-0661-9
8. Celik IH, Demirel G, Uras N, Oguz ES, Erdeve O, Dilmen U. [The role of serum interleukin-6 and C-reactive protein levels for differentiating aetiol- ogy of neonatal sepsis]. Arch Argent Pediatr 2015;
113(6):534-7. DOI: 10.5546/aap.2015.534
9. Khaertynov KS, Boichuk SV, Khaiboullina SF, Anokhin VA, Andreeva AA, Lombardi VC, et al. Comparative Assessment of Cytokine Pattern in Early and Late Onset of Neonatal Sep - sis. J Immunol Res 2017; 2017:8601063. DOI:
10.1155/2017/8601063
10. Steinberger E, Hofer N, Resch B. Cord blood procalcitonin and Interleukin-6 are high- ly sensitive and specific in the prediction of early-onset sepsis in preterm infants. Scand J Clin Lab Invest 2014; 74(5):432-6. DOI:
10.3109/00365513.2014.900696
11. Celik IH, Demirel FG, Uras N, Oguz SS, Erdeve O, Biyikli Z, Dilmen U. What are the cut-off levels for IL-6 and CRP in neonatal sepsis?. J Clin Lab Anal 2010; 24(6):407-12. DOI: 10.1002/jcla.20420
12. Boskabadi H, Zakerihamidi M. Evaluate the diagno- sis of neonatal sepsis by measuring interleukins: A systematic review. Pediatr Neonatol 2018; 59(4):
329-338. DOI: 10.1016/j.pedneo.2017.10.004
13. Machado JR, Soave DF, da Silva MV, de Menez- es LB, Etchebehere RM, Monteiro ML, et al. Neonatal sepsis and inflammatory mediators. Mediators Inflamm 2014; 2014:269681. DOI: 10.1155/2014/269681
14. Mirzarahimi M, Barak M, Eslami A, Enteshari-Mogh- addam A. The role of interleukin-6 in the early diagnosis of sepsis in premature infants. Pediatr Rep 2017; 9(3):7305. DOI: 10.4081/pr.2017.7305
13. Barug D, Goorden S, Herruer M, Müller M, Brohet R, de Winter P. Reference values for interleukin-6 and interleukin-8 in cord blood of healthy term neonates and their association with stress-re- lated perinatal factors. PLoS One 2014; 9(12): e114109. DOI: 10.1371/journal.pone.0114109
16. Ye Q, Du LZ, Shao WX, Shang SQ. Utility of cy- tokines to predict neonatal sepsis. Pediatr Res 2017; 81(4):616-621. DOI: 10.1038/pr.2016.267.E
17. Chauhan N, Tiwari S, Jain U. Potential biomark- ers for effective screening of neonatal sepsis infections: An overview. Microb Pathog 2017; 107:234-242. DOI: 10.1016/j.micpath.2017.03.042
18. Mirzarahimi M, Barak M, Eslami A, Enteshari-Mogh- addam A. The role of interleukin-6 in the early diagnosis of sepsis in premature infants. Pediatr Rep 2017; 9(3): 7305. DOI: 10.4081/pr.2017.7305
19. Chiesa C, Pacifico L, Natale F, Hofer N, Osborn JF, Resch B. Fetal and early neonatal interleu- kin-6 response. Cytokine. 2015; 76(1):1-12. DOI: 10.1016/j.cyto.2015.03.015
20. Boskabadi H, Zakerihamidi M. Evaluate the diagno- sis of neonatal sepsis by measuring interleukins: A systematic review. Pediatr Neonatol 2018; 59(4): 329-338. DOI: 10.1016/j.pedneo.2017.10.004
21. Sorokin Y, Romero R, Mele L, Iams JD, Peace- man AM, Leveno KJ, et al. Umbilical cord serum interleukin-6, C-reactive protein, and myeloper- oxidase concentrations at birth and association with neonatal morbidities and long-term neuro- developmental outcomes. Am J Perinatol 2014; 31(8):717-26. DOI: 10.1055/s-0033-1359723
22. Alves JB, Gabani FL, Ferrari RAP, Tacla MTGM, Linck Júnior A. Neonatal sepsis: mortality in a municipality in southern Brazil, 2000 to 2013. Rev Paul Pediatr 2018; 36(2): 132-140. DOI: 10.1590/1984-0462/;2018;36;2;00001Molloy EJ, O’Neill SM. Neonatal Escherichia coli sepsis: possible effects of gender, type of surfactant, or time to initiation of antibiotics?. Am J Obstet Gynecol 2006; 194(4):1201-2. DOI: 10.1016/j.ajog.2005.07.088
23. Kurt AN, Aygun AD, Godekmerdan A, Kurt A, Dogan Y, Yilmaz E. Serum IL-1beta,IL-6, IL-8, and TNF-al- ha levels in early diagnosis and management of neonatal sepsis. Mediators Inflamm 2007; 2007: 31397. DOI: 10.1155/2007/31397
24. Bhandari V. Effective Biomarkers for Diagnosis of Neonatal Sepsis. J Pediatric Infect Dis Soc 2014; 3(3):234-45. DOI: 10.1093/jpids/piu06325. Sugitharini V, Prema A, Berla Thangam E. Inflam- matory mediators of systemic inflammation in neonatal sepsis. Inflamm Res 2013; 62(12):1025-34. DOI: 10.1007/s00011-013-0661-9
-
26. Krauss-Silva L, Moreira ME, Alves MB, Rezende MR, Braga A, Camacho KG, et al. Randomized controlled trial of probiotics for the prevention of spontaneous preterm delivery associated with intrauterine infection: study protocol. Reprod Health 2010; 7:14. DOI: 10.1186/1742-4755-7-14
27. Pavare J, Grope I, Kalnins I, Gardovska D. High- mobility group box-1 protein, lipopolysacchari- de-binding protein, interleukin-6 and C-reactive protein in children with community acquired infections and bacteraemia: a prospective study. BMC Infect Dis 2010; 10:28. DOI: 10.1186/1471- 2334-10-28
![]()