 Artículo de Investigación
Artículo de Investigación Artículo de Investigación
Artículo de InvestigaciónFabricio González-Andrade1, Carlos Torres-Serrano2, Jakeline Pinos3, María del Carmen Grijalva4, Gabriela Aguinaga-Romero5
1. MD, PhD. Universidad Central del Ecuador, Facultad de Ciencias Médicas, Unidad de Medicina Traslacional, Iquique N14-121 y Sodiro-Itchimbía, 170403, Quito, Ecuador. Universidad San Francisco de Quito USFQ, Colegio Ciencias de la Salud, calle Diego de Robles s/n y Pampite, 170901, Quito, Ecuador. ORCID: 0000-0002-2091-9095. E-mail: fabriciogonzaleza@gmail.com. Corresponding autor.
2 MD, MSc. Universidad Central del Ecuador, Facultad de Ciencias Médicas, Unidad de Medicina Traslacional, Iquique N14-121 y Sodiro-Itchimbía, 170403, Quito, Ecuador. ORCID: 0000-0002-2695-7877. E-mail: ctorresserrano@gmail.com.
3 MD. Programa Ampliado de Inmunizaciones, Ministerio de Salud Pública, Av. Quitumbe Ñan y Av. Amaru Ñan, Plataforma Gubernamental de Desarrollo Social, 170146, Quito, Ecuador. ORCID: 0000-0002-6820-1541. E-mail: jakepinos@gmail.com.
4 MD. Programa Ampliado de Inmunizaciones, Ministerio de Salud Pública, Av. Quitumbe Ñan y Av. Amaru Ñan, Plataforma Gubernamental de Desarrollo Social, 170146, Quito, Ecuador. ORCID: 0000-0001-5769-7484. E-mail: mcgrijalva@hotmail.com.
5 MD, MPH. Universidad Central del Ecuador, Facultad de Ciencias Médicas, Unidad de Medicina Traslacional, Iquique N14-121 y Sodiro-Itchimbía, 170403, Quito, Ecuador. ORCID: 0000-0002-6274-2195. E-mail: gabrielaaguinaga@yahoo.com
Archivos de Medicina (Manizales) Volumen 20 N° 1, Enero-Junio 2020, ISSN versión impresa 1657-320X, ISSN versión en línea 2339-3874, González Andrade F., Torres Serrano C., Pinos J., Grijalva M.dC., Aguinaga Romero G.
Recibido para publicación: 17-04-2019 - Versión corregida: 20-11-2019 - Aprobado para publicación: 27-11-2019
González-Andrade F, Torres-Serrano C, Pinos J, Grijalva MdC, Aguinaga-Romero G. Diagnostic screening of HPV genotypes in 555 Ecuadorian mestizo women of seven provinces, and comparison with other Latino American populations. Arch Med (Manizales) 2020; 20(1):86-96.DOI: hhttps://doi.org/10.30554/archmed.20.1.3296.2020
Objective: this paper aims to perform diagnostic screening of HPV in healthy Ecuadorian mestizo women, from seven provinces, and compare the findings with other Latin American populations. Material and methods: genotyping was done with two different oligonucleotides MY09 and MY11; a fragment of 450 base pairs was amplified, L1 region of the viral genome. Results: it analyzed 555 women, 35 were positive for HPV (6.3%). Genotypes found in relation to oncogenic risk, were 6, 11, 16, 18, 31, 33, 35, 42, 45, 51, 52, 53, 58, 59, 61, 81. 12/35 women (34.3%) presented high-risk genotypes. Four positive cases were also observed in women older than 55 years (0.36%). The 14 published studies of Ecuadorian women showed that the most prevalent genotypes are 16, 18, 31, 52, 53, 56 and 58; while in the eight Latin American Studies the most prevalent are 16, 18, 31, 45, 52 and 58. Conclusion: although there are several studies on HPV genotyping on Latin American populations, there is an important gap related to ethnicity and the prevalence of the virus. In addition, most of them have not compared similar and common subtypes in the population. The general prevalence of HPV in the studied population was 6.3%. It found that genotypes 16, 18, 31, 52, 53, 56 and 58 are the most prevalent in Ecuadorian normal mestizo women. Genotypes 53 and 56 are common in Latino populations. Larger studies, in different ethnic groups are needed to identify other prevalent genotypes in certain geographical areas.
Keywords: Papillomavirus Infections, diagnosis, Ecuador, mass screening, health planning.
Objetivo: realizar tamizaje diagnóstico de VPH en mujeres mestizas ecuatorianas sanas, de siete provincias, y comparar los hallazgos con otras poblaciones latinoamericanas. Material y métodos: el genotipado se realizó con dos oligonucleótidos diferentes MY٠٩ y MY١١; se amplificó un fragmento de 450 pares de bases, correspondiente a la región L1 del genoma viral. Resultados: se analizó 555 mujeres, 35 fueron positivas para VPH (6,3%). Los genotipos encontrados en relación con el riesgo oncogénico fueron 6, 11, 16, 18, 31, 33, 35, 42, 45, 51, 52, 53, 58, 59, 61, 81. 12/35 mujeres (34,3%) presentaron genotipos de alto riesgo. También se observaron cuatro casos positivos en mujeres mayores de 55 años (0,36%). Los 14 estudios publicados de mujeres ecuatorianas mostraron que los genotipos más prevalentes son 16, 18, 31, 52, 53, 56 y 58; mientras que en los ocho estudios latinoamericanos los más prevalentes son 16, 18, 31, 45, 52 y 58. Conclusión: aunque existen estudios sobre la prevalencia de VPH, aún existe una brecha sobre la relación entre la etnicidad y la prevalencia del virus. Además, los estudios en América Latina no han comparado subtipos similares y comunes en la población. La prevalencia general del VPH en la población estudiada fue del 6,3%. Este estudio encontró que los genotipos 16, 18, 31, 52, 53, 56 y 58 son los más prevalentes en las mujeres mestizas ecuatorianas sanas. Los genotipos 53 y 56 son los más comunes en las poblaciones latinas. Se necesitan estudios más grandes, en diferentes grupos étnicos para identificar otros genotipos prevalentes en ciertas áreas geográficas.
Palabras clave: infecciones por el virus del papiloma, diagnóstico, Ecuador, detección masiva, planificación de la salud.
Although there are several studies on HPV genotyping on Latin American populations, there is an important gap related to ethnicity and the prevalence of the virus in these populations. In addition, most of them have not compared similar and common subtypes between populations, so it cannot always generalize the data about the prevalence of viruses. In addition, although subtypes of high pathogenicity have already been established, there are still circumstances to be characterized according to each population and the behavior of the virus in each of them.
The human papilloma virus (HPV) is the causative agent of cervical cancer [1,2]. This heterogeneous virus’s family includes more than 200 genotypes; among which, more than 40 types of HPV can be easily spread through the genital tract. Fourteen HPV genotypes (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66 and 68) are considered pathogenic or high risk to cause cervical cancer [1]. Although most sexually active women become infected with HPV at least once in their lifetime, less than 10% of women are persistently infected, and it is the persistent infection with a high-risk HPV genotype that contributes to the development of cervical cancer. It has also been shown that majority of HPV infections are self-limiting, and can cure almost completely [2].
Virtually all cervical neoplasms are caused by HPV [3,4]. HPV accounts for 40% of vulvar cancers, 70% of vaginal cancers, 40% of penile cancers, and 85% of anal cancer [3]. High-risk genotypes can cause intraepithelial neoplasia of the urogenital region, including cervix, vulva, vagina, penis, anal and cancer, as well as some cancers of the oropharynx [3]. There are identified more than 30 genotypes that infect the urogenital region, and at least 10 of them are directly associated with intracervical neoplasia [4,5].
HPV belongs to the papillomavirus family, and its classification is clinically significant for the following reasons: 1) not only one specific genotype of HPV is associated with cervical cancer; 2) the pathogenicity of HPV varies according to the genotype. HPV is grouped into five genera (alpha, beta, gamma, mu and nu), and the genus alpha papillomavirus includes HPV genotypes that infect both oral and genital mucosa. In addition, HPV can be classified according to its L1 genome sequence, type, intratypic lineage and sub lineage. The different types, intra-typic lineages and sublines of HPV are defined as having L1 sequences that differ by at least 10 ± 1%, respectively [4,5].
HPV can also be grouped into high risk and low risk categories based on their oncogenic potential. Among the 14 high-risk HPV genotypes (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, 59, 66, and 68), the two most common (16 and 18) are the factors that cause 71% of cervical cancers, in developed countries. Two genotypes of low risk HPV (6 and 11) contribute to the formation of genital warts, most of which require treatment [6,7].
As the guidelines for cervical cancer screening continue to evolve, HPV genotyping has assumed a significant role in the recently published recommendations [8]. In 2014, the FDA approved an HPV test for use in the primary detection of cervical cancer. In addition, the Society of Gynecologic Oncology and the American Society of Colposcopy and Cervical Pathology have published provisional guidelines that supported screening with a single HPV test, without cytology, every 3 years for women ≥25 years [9,10]. On the basis of previous data, the guidelines recommend the use of different clinical approaches for the management of patients infected with different genotypes of HPV9. Positive patients for HPV 16/18 should undergo a colposcopy, while patients infected with any of the other 12 high-risk HPV types will undergo a liquid cytology test. An HPV test is more sensitive than cytology to detect cervical precancerous cervical lesions [11,12].
On the other hand, prophylactic vaccines against HPV have been developed based on recombinant expressed virus-like particles. Two first-generation vaccines approved by the FDA prevent infections and diseases caused by HPV 16 and 18, the two HPV genotypes that cause approximately 70% of cervical cancer, and one of these vaccines also prevents HPV 6 and 11 , the two most frequent HPV genotypes that cause 90% of genital warts. A new generation vaccine, recently approved by the FDA, prevents HPV 16, 18 and five additional HPV genotypes that together cause approximately 90% of cervical cancer in Caucasian populations [12]. In clinical trials, these vaccines have shown high levels of efficacy against diseases and infections caused by specific genotypes of HPV in adolescent women and men and older women. The data indicate that the effectiveness of the population, and therefore the cost-effectiveness ratio, is higher in young women before they become sexually active [12].
The objective of this study is to show the prevalence of HPV genotypes in Ecuadorian mestizo women [13], in comparison with other studies at Latin America [14-33] .
Study design: epidemiological, observational, cross-sectional study, with one cohort of patients. After that, it makes a simple comparison with other related populations.
Settings: outpatient gynecology services at Ministry of Health (MoH) facilities in five cities of the country, from the provinces of Guayas, Pichincha, El Oro, Esmeraldas, Sucumbíos, Bolívar and Napo. The samples were collected between January 2016 and December 2017.
Participants: Ecuadorian mestizo women, born and living in Ecuador, between 18 and 68 years of age. All women included were randomly selected, were healthy, and not show any previous report of HPV suspicion.
Study size: 555 women
Data sources: personal interview, sampling and clinical records of each patient.
Bias avoidance: data collected by the same person always. It performed laboratory test by the same professional in all cases.
Genotyping: pre-analytical phase, each sample of uterine cervix was collected by brushing the endo and exo cervix, which was transported in a solution of phosphate buffer and cold chain to the university laboratory. PCR analytical phase, DNA extraction from cervical brushes was performed using the QIAamp DNA Mini Kit (Qiagen Ltd, Crawley, United Kingdom), according to the manufacturer’s instructions. Aliquots of 200 μl of digested samples were taken with 20 μl of proteinase K and 200 μl of buffer at 56 ° C, for 10 minutes. The precipitation of the DNA was carried out by the addition of 200 μl of 96% ethanol. The DNA was eluted in 200 μl of buffer and stored at 20° C until further use. The detection of the virus was done with two different oligonucleotides MY09 (5’CGTCCMARRGGAWACTGATC3’) and MY11 (5’GCMCAGGGWCATAAYAATGG3’). A fragment of 450 base pairs was amplified, corresponding to the L1 region of the viral genome. The PCR reactions for each oligonucleotide primer were carried out separately, for each PCR reaction, a positive control was used, DNA extracted from a condyloma sample, and a negative control, H2O. The result was considered positive for DNA-HPV when one of the oligonucleotide primer test MY09/MY11 detected the viral DNA. The PCR protocol used 15 mM MgCl2 (1 × buffer), 800 μM of dNTPs, 50 pmol/μl of each oligonucleotide, 1.25 U of Taq DNA polymerase hot star; the thermocycle used 40 cycles at 94°C/45 seconds, 55°C/45 seconds, 72°C/45 seconds, 1 cycle of 72°C/ 5 min. Post-PCR phase: the analysis of the PCR products was carried out using RFLPs using seven different endonucleases; the reading was made on agarose gels, with electrophoresis and staining with ethidium bromide.
Statistical analysis: Data collected from the clinical records; it analyzed with the statistical program SPSS v23.0. Frequencies, percentages and correlations were determined.
Institutional Board Authorization
All the patients signed the informed consent according to the Declaration of Helsinki. The Institutional Review Board of the Universidad Central del Ecuador approved the research protocol before the study began.
The average age of the patients was 31.29±9.4 years, with a range of 14 to 68 years. According with the age, 129 patients (23.2%) of 25 to 29 years show highest prevalence of HPV, followed by the group of 20 to 24 years with 97 patients (17.5%), and the group of 30 to 34 years with 87 women (15.7%). Women in childbearing age were the 56.4%. The majority of patients came from the province of Pichincha with 105 women (18.9%) followed by El Oro with 104 women (18.7%).
The average age of onset of active sexual life of the women studied were 17.79±3.43 years, with a range of 10 to 34 years. The average number of sexual relations that the patients maintained was 2.32±1.07 per week, the mean found was one sexual contact per week. The average of sexual partners was 2±0.93, with a range of 1 to 25 sexual partners, with a single partner in 50.6% of the patients. The time of contraceptive use was an average of 35.19±13.18 months, with a range of 0 to 180 months, which shows that the majority of women, 41.2%, do not use contraceptives.
Table 1 shows the distribution of genotypes found in relation to oncogenic risk, with genotypes 6,11,16,18,31,33,35,42,45,51,52,53,58,59,61,81. This table also shows the distribution of findings found according to age group.
Table 1. Distribution of findings found according to age group, and distribution of genotypes found in relation to oncogenic risk
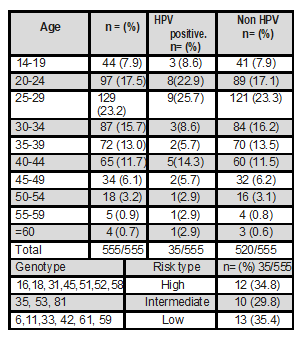
Source: study data. Elaboration: authors.
Table 2 makes a comparison of the findings found in comparison with other studies published at national and Latin American level. It shows patients with a single genotype and with multiple genotypes.
Table 2. Comparison of the findings found in comparison with other studies published at national and Latin American level. It shows patients with a single genotype and with multiple genotypes
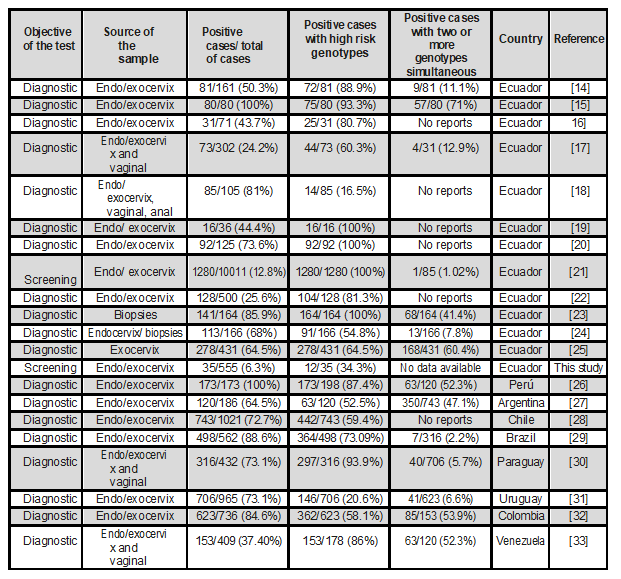
Source: study data. Elaboration: authors.
Finally, Table 3 shows the comparison of the genotypes found in this study versus the genotypes found in studies conducted in other countries.
Table 3. Comparison of the genotypes found in this study versus the genotypes found in studies conducted at the national and Latin American level (LATAM)
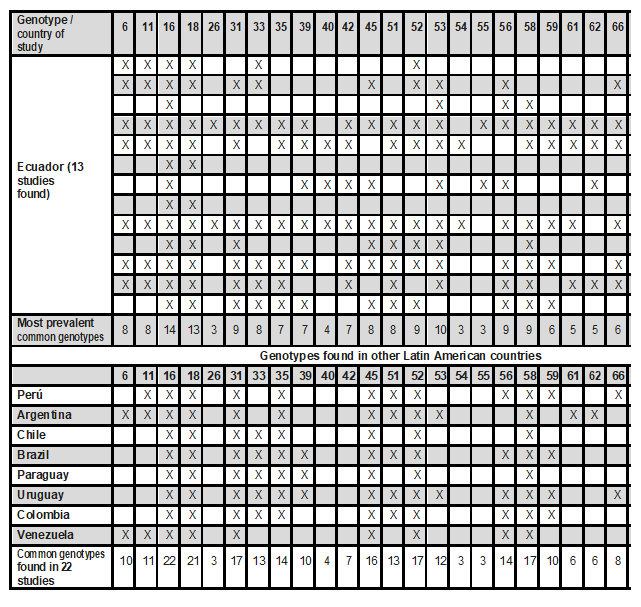
Four studies conducted in Ecuador show unique genotypes such as 32, 43, 44, 64, 67, 72, 73, 82, 83, 84. * = this study. Source: study data. Elaboration: authors.
Although the scientific literature shows clear evidence that DNA tests for HPV are more sensitive to identify women with CIN compared to standard cytology, since the latter has a lower specificity, Ecuador does not have a Cervical Cancer Screening Program based on the detection of HPV, only the primary detection by Pap test is mandatory. For this reason, the data that exists are research studies conducted in isolation, and oriented to health care services. New screening strategies for cervical cancer detection support the use of the HPV test as a primary test, due to its greater sensitivity, followed by cytology in those patients with positive HPV results; this is due to the greater specificity of cytology [34].
The Ministry of Health of Ecuador, within the National Cancer Strategy 2013 to 2023 has arranged to perform screening in women aged 21 to 65 with Pap smear every 3 years. In women aged 30 to 65 years, perform screening with cytology and molecular tests for HPV DNA every 5 years, the latter is not mandatory. This contrasts with the fact that in 2015 the mortality from cervical cancer in Ecuadorian women was 8.4% of the total cancer cases, being the third leading cause of death in women, in our country in that year. Health authorities in Ecuador suggest that in patients aged 30 to 65, with a positive Pap test, the DNA test for HPV is requested, and if the latter is positive, a colposcopy is additionally requested. Although it seems an appropriate strategy, it has not been fully implemented in Ecuador.
The demographic profile of the patients in this study was mostly women between 20 and 34 years old, most of them from the provinces of Pichincha and El Oro, who came to control in the gynecological outpatient clinic. Pichincha province shows greater amount of patients because the healthcare centers of attention at national level are located in this city. The majority of patients came from the province of Pichincha with 105 women (18.9%) followed by El Oro with 104 women (18.7%).
The average age of the patients was 31.29±9.4 years, with a range of 14 to 68 years. It is common for the medical consultation of health facilities in Ecuador to attend women of all ages. The patient with the lowest positive for HPV was 14 years old; this is related to the high rates of early onset of sexual activity and pregnancy in adolescents in our country. This contrasts with the lowest age found in a similar study performed in a European country such as Portugal [35], where the minimum age was 25 years. This is explained by the patterns of sexual behavior that differ between the two countries. Of course there is more sex education in Europe.
According with the age, the group between 25 to 29 years, shows highest prevalence of HPV. This data is similar in all countries. Women in childbearing age were the 56.4%. The mean age of onset of active sexual life of the women studied were 17.79±3.43 years, with a range of 10 to 34 years. The mean number of sexual relations that the patients maintained was 2.32±1.07 per week, the mean found was one sexual contact per week. The average of sexual partners was 2±0.93, with a range of 1 to 25 sexual partners, with a single partner in 50.6% of the patients. The time of contraceptive use was an average of 35.19±13.18 months, with a range of 0 to 180 months, which shows that the majority of women, 41.2%, do not use contraceptives.
Of a total of 555 women, 35 were positive for HPV, which represents 6.3%. Of these, 34.3% showed high-risk genotypes (12/35). Four positive cases were also observed in women older than 55 years, equivalent to 0.36% of the analyzed sample. The general prevalence of HPV in the studied population was 6.3%; this percentage is lower in comparison with other countries as Mexico that reports in healthy women only 9.1% [36], Turkey with 17.9% [37], or East China with 10.6% [38]. It is important to note that there are very few diagnostic screening studies conducted in healthy women. Comparing the studies published in Latin America and Ecuador, only two papers on diagnostic screening were observed and the rest are on the diagnostic confirmation of HPV. Screening diagnostic studies are rare, although, they should be done to understand the local epidemiology of the disease.
Comparison with other populations
Firstly, it is important to note that there are very few diagnostic screening studies conducted in healthy women. Comparing the studies published in Latin America and Ecuador, only two papers on diagnostic screening were observed and the rest are on the diagnostic confirmation of HPV. Screening diagnostic studies are rare, although, they should be done to understand the local epidemiology of the disease.
Firstly, there is no homogeneity in the origin of the tissues of the samples; although most studies analyzed samples of endo and exo-uterine cervix. In six studies were also included vaginal and anal biopsies samples.
The published studies of Ecuadorian women showed that the most prevalent genotypes are 16, 18, 31, 52, 53, 56 and 58; while in the eight Latin American studies the most prevalent are 16, 18, 31, 45, 52 and 58. In both, regional groups, genotypes 16, 18 and 31 are most common. However, there are two different genotypes between them. It is possible that it is due to ethnic variations and population substructure typical of each country of origin. All the women analyzed in this study were mestizas, and in some admixtures studies, miscegenation is mentioned as a protective factor against certain diseases. Miscegenation by definition refers to the encounter between populations and their interbreeding. Genetic biodiversity has a population, anthropological, migratory, forensic and medical interest. All Ecuadorians come from a mixture of three original groups: Caucasian Europeans, Native Amerindians and Africans. More studies are required to explain this phenomenon.
Most cases of HPV-associated cancer are caused by HPV genotypes 16 and 18, which account for 66% of cervical cancer cases in the USA, and genotypes 31, 33, 45, 52 and 58 of HPV. They represent an additional 15% of cases of cervical cancer. For cervical intraepithelial neoplasia, 50-60% of cases are caused by genotypes 16 and 18 of HPV, and 25% of cases are caused by genotypes 31, 33, 45, 52 and 58 of HPV. Roughly 90% of cases of genital warts are caused by genotypes 6 and 11 of HPV. That is, in theory, the vaccine provides protection for 81% of the most frequent genotypes that cause cervical cancer and 90% of the condylomas.
In different studies, HPV53 has been considered and/or classified as a medium-high risk virus, although isolated infection and the development of dysplastic lesions have been little studied. More studies and comparative monitoring are needed in other socio-geographical areas to establish the true risk. Genotype 56 has high evidence to be considered high risk. It is important to note that four studies conducted in Ecuador show unique genotypes such as 32, 43, 44, 64, 67, 72, 73, 82, 83, and 84, observed in a single study.
Limitations of the study
This work is very important in this knowledge field, although it has three limitations: first, the sample, although important, is still insufficient. This is because there were limited resources for laboratory analysis. Second, the coverage of the sample is incomplete; samples from all provinces were missing. Unfortunately, there were logistical limitations when it came to capturing patients. Third, the samples were limited to MoH health facilities, leaving other health institutions in the country without participating in the study. Future research should expand the number of samples, improve sampling, achieve national coverage, be multicenter and achieve the participation of all actors in the health system.
Conclusion
Although there are several studies on genotyping of HPV there is a gap about the ethnicity and prevalence of the virus. In addition, studies in Latin America have not compared similar and common subtypes in the population. The general prevalence of HPV in the studied population was 6.3%. It found that genotypes 16, 18, 31, 52, 53, 56 and 58 are the most prevalent in Ecuadorian normal mestizo women. Genotypes 53 and 56 are common in Latino populations. Larger studies, in different ethnic groups are needed to identify other prevalent genotypes in certain geographical areas.
Author’s Contributions
The research protocol and its design; data collection, statistical analysis, evaluation and interpretation of the data, critical analysis, discussion, writing and approval of the final manuscript were carried out by all authors. They contributed in the same way in the whole process. The corresponding author represents the group of authors.
Availability of Data and Materials
The data supporting this manuscript are available upon request to the corresponding author.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest: the authors report NO conflict of interest
Sources of funding: the author declares that the financial resources for the elaboration of this research do not come from any external funds. This research was self-funded.
