 Artículo de Investigación
Artículo de Investigación 
Cellular count changes in different rat brain areas due to early maternal separation
Alejandro Muñoz Valencia1, Jonatan Velásquez Quiroga 2, Javier Saddam López Arias 3, Jhon Chavarro Tibacan 4, Zulma Dueñas 5
1 Medical Student. Universidad Nacional de Colombia. Bogotá. Colombia. ORCID: 0000-0001-6845-5130. Correo e.: alemunozvalunal.edu.co.
2 Urology Resident. Universidad Nacional de Colombia. Bogotá. Colombia. Correo e.: javelasquezq@unal.edu.co.
3 Medical Doctor. Universidad Nacional de Colombia. Bogotá. Colombia. Correo e.: jaslopezar@unal.edu.co.
4 Pharmacy Student. Universidad Nacional de Colombia. Bogotá. Colombia. Correo e.: jachavarrot@unal.edu.co.
5 Professor. Facultad de Medicina. Departamento de Ciencias Fisiológicas. Universidad Nacional de Colombia. Bogotá. Colombia. ORCID: 0000-0001-6068-4174. Correo e.:zjduenasg@unal.edu.co. Corresponding author.
Archivos de Medicina (Manizales), Volumen 19 N° 1, Enero-Junio 2019, ISSN versión impresa 1657-320X, ISSN versión en línea 2339-3874. Muñoz Valencia A.; Velásquez Quiroga J.; López Arias J.S.; Chavarro Tibacan J.; Dueñas Z.
Recibido para publicación: 14-12-2018 - Versión corregida: 28-02-2019 - Aprobado para publicación: 06-03-2019
Muñoz-Valencia A, Velásquez-Quiroga J, López-Arias JS, Chavarro-Tibacan J, Dueñas Z. Cellular count in motor cortex and maternal separation. Arch Med (Manizales) 2019; 19(1):12-22. DOI: https://doi.org/10.30554/archmed.19.1.2983.2019.
Summary
Objective: to identify whether maternal separation during breastfeeding (MSDB) affects the cellular count in different rat brain areas. The continuous mother-child interaction, adjusts and modulates the offspring behavioral response to environmental stimuli and also affects their development and homeostasis. Morphological and physiological changes in the offspring brains have been observed, including cell count changes in different brain areas with differences between males and females. Materials and methods: this study compared albino Wistar rats in a protocol of MSDB with a control group. Brain tissue was fixed with paraformaldehyde, cut in cryostat and either stained with Hematoxylin-Eosin (H&E) or processed for immunohistochemistry against glial fibrillary acidic protein (GFAP). All sections were analyzed using a cell count protocol including statistical analysis with Students T test at a significance level of P ≤0.05. Results: the MSDB group of male subjects presented higher GFAP-marked cell count in primary motor cortex and hippocampus; while female subjects, showed less GFAP-marked cell count in these same areas. Conclusions: MSDB produces sex-specific changes in the number of glial cells especially in the primary motor cortex, this finding may be considered as associated factor of alterations in motor responses to stress in these subjects, in addition to other known causes such as the Hypothalamic-Pituitary-Adrenal Axis dysfunction.
Key Words: breast feeding, motor cortex, hippocampus, child development.
Cambios en el recuento celular en diferentes áreas del cerebro de rata debido a la separación materna temprana
Resumen
Objetivo: identificar si la separación materna durante la lactancia (MSDB) afecta el conteo celular en cortezas motoras y otras áreas del cerebro de la rata. La interacción continua entre la madre y su hijo, ajusta y modula la respuesta comportamental de las crías hacia estímulos ambientales, afectando además su desarrollo y homeostasis. Cambios morfológicos y fisiológicos han sido descritos en la descendencia, incluyendo diferencias en conteos celulares de varias áreas de la corteza cerebral. Materiales y métodos: este estudio se compararon ratas Albino Wistar bajo un protocolo de MSDB contra un grupo control. El tejido cerebral fue fijado con paraformaldehido, cortado en criostato, y tratado con tinción Hematoxilina-Eosina (H&E) o procesado con inmunohistoquimica contra proteína acida glíal fibrilar (GFAP). Todas las secciones fueron analizadas usando un protocolo de conteo que incluyo análisis estadístico con el test de T de Student con significancia a nivel de P ≤0.05. Resultados: el grupo de machos que tuvo MSDB presentó mayor conteo de células marcadas contra GFAP en las cortezas motoras primarias y el hipocampo; mientras que las hembras con MSDB, mostraron menor conteo de células marcadas contra GFAP en estas mismas áreas. Conclusión: la MSDB produce cambios específicos de acuerdo al sexo del sujeto, en el número de células gliales especialmente en las cortezas motoras primarias, este hallazgo puede ser considerado como causante parcial de las alteraciones en las respuestas motoras a estrés en estos sujetos, además de otras causas conocidas como la disfunción del eje Hipotálamo-Pituitario-Adrenal (HPA).
Palabras clave: lactancia materna, corteza motora, hipocampo, desarrollo infantil.
Introduction
Epidemiological evidence in humans associates early exposure to adverse events with greater vulnerability to develop mental diseases such as anxiety attacks, depression, substance abuse and behavioral changes, among others, including subsequent social misbehavior [1-4]. Such evidence has allowed to postulate that the acquired mental dysfunctions may depend on alterations of the neuronal circuits and failure to accomplish different developmental mechanisms. With this background, it is important to take into account that maternal bond is fundamental to establish synaptic networks and also plays a crucial role in the postnatal development of individuals [5-9]. Observational studies that can be done with human subjects are not able to show the underlying mechanisms that influence the offspring in terms of stress response and neuro-histological correlation. Thus, experimental methods with other mammal species as animal models allow different approaches to this issue. Most researches has been done with rodents, especially rats, for which the widest and most detailed knowledge exists [10-13].
The maternal separation during breastfeeding (MSDB) animal protocol in rats has revealed numerous anatomical and physiological changes in the hypothalamus-pituitary-adrenal axis (HPA), amygdala, hippocampus, prefrontal and medial cortex, and peri hypothalamic areas such as the medial preoptic area [14-16]. The GABAergic system, closely related to the regulation of anxiety, is one of those most affected by MSDB, especially the expression of GABAA receptors in the hippocampus [17] and medial prefrontal cortex [18]. However, there are few references regarding cellular changes in the amygdala, hypothalamus and peri hypothalamic area [18], which are important in the regulation of responses to fear and anxiety [19].
Furthermore, the response to stress and anxiety is strongly mediated by factors related to the sex of the animals [16]. Few studies using MSDB that have explored the neurobiological and behavioral effects, had grouped or analyzed according to each sex group [16-18]. Previous studies have reported that exposure to a protocol of MSDB causes differences in anxiety-like behaviors, which seem to depend on subject’s sex and also can be associated with the alteration of expression by GABAA receptors. Other authors support evidence regarding the number of astrocytes in certain brain areas of separated rats compared with those that were not separated [18, 20, 21].
The maternal separation has also been linked to postnatal maturation of neural cells, specifically of microglial cells in various areas of the central nervous system [22]. Other associations were observed between the maternal separation and lower gene expression of GFAP in glial cells at the prefrontal cortex at different postnatal stages, including adult rats up to 10 weeks old [23]. Morphometric abnormalities of the ventricles seen in rodent models have been associated with different pathologies including substance abuse disorders and Alzheimer’s disease [24,25].
Taking in to account all these antecedents, the objective of the present study was to identify the possible changes induced by MSDB in the total cell counting of GFAP and non-GFAP cells, along different areas including motor cortices of the rat brain and to describe whether these changes were different between male and female groups, comparing all data with control groups.
Materials and methods
Maternal Separation During Breastfeeding
Female and male Wistar rats were obtained from the in-house animal facility of the Universidad Nacional de Colombia and housed at the veterinary school. Animals were housed in standard rat cages (40 x 31 x 22 cm) and kept under standard laboratory conditions with reversed light/dark cycle (lights off at 07:00 h), 22 ± 2°C temperature, 55 ± 10% humidity and food and water ad libitum. Animals were mated during 1 week; and housed alone during the last 2 weeks of gestation. Litters were randomly assigned to separation or animal facility-reared (control) groups to equally distribute each gender. All experimental protocols were approved by the Ethics Committee of the Facultad de Medicina de la Universidad Nacional de Colombia and according to Colombian law (Resolución N°. 8430 de 1993) and international regulations regarding research using animal subjects i.e. National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications) and in accordance with the rules and procedures of the APA.
Ethics Committee. Four dams were used. Litters were undisturbed at birth: postnatal day zero (P0) and were homogenized to 10. Four groups were set, two with only males and two with only females, setting one control and one experimental group for each sex, respectively. The MSDB protocol was the same as reported in previous studies [18]. From day 0 to day 22, the protocol for experimental groups comprised 180 minutes of separation from the mother during the morning and 180 minutes during the afternoon. Then, on day 22 all subjects were separated from the mother, and four group were set by subject’ sex and previous exposure to MSDB protocol or control, setting up each group with 10 subjects procuring equal male: female ratios. Conditions of illumination, temperature, food, and drink were equal for all subjects. The rats continued their growing and development with habituation to minimal human handling, including the change of bedding and food supply until maturation. On day 60, the subjects were anesthetized, then, transcardial perfusion was carried out with saline solution 0.9% and then 4% paraformaldehyde (PFA). The brains were preserved in PFA for further histological analysis.
Histological processing for cellular identification
The brains were cryoprotected in a saturated sucrose solution four days before sectioning in cryostat. The brains were later processed by carrying out a series of coronal sections of 20 μm thick. Half of the slices were stained with H&E to identify the total number of cells in each of the structures and the remaining half was stained for immunohistochemistry against GFAP.
Histological processing for immunohistochemistry
The procedure was performed as previously reported [20]. The 20μm slices were incubated with blocking serum [bovine serum albumin, 0.3% Triton X-100, normal goat serum] for 1 h. Then, incubated with primary antibody anti-GFAP (Sigma-Aldrich Cat# G4546, RRID:AB_1840895) for 12h at room temperature before undergo incubation with biotinylated anti-rabbit secondary antibody (Vector Laboratories Cat# PK-6100, RRID:AB_2336819) at a dilution of 1:500 in 0.3 % PBS Triton for 90 min, and then with antibody (AB) complex for 90 min. Finally, the slices were treated with Diaminobenzidine (Vector Laboratories Cat# SK-4100, RRID:AB_2336382). Four rinses with 0.3% PBS Triton were carried out for 15 minutes between each stage. After clearing the DAB and adding 0.9% NaCl solution, the slices were mounted on slides with a fine paintbrush for later analyses.
Cell count and statistical analysis
Photographic records were taken of each sections using the 2X and 4X objectives of a CARL ZEISS inverted microscope linked to a Cannon® G10 digital camera, connected to a computer where the images were stored. Three photos of each brain area of interest were taken from each subject; the photos were sorted by region, staining and sex.
Analyzed areas were identified according to the stereotaxic coordinates described by Paxinos & Watson [26] as follows: Area 1 including M1 – primary motor cortex, LO – lateral orbital cortex, and VO – ventral orbital cortex at Interaural 14.20 mm and Bregma 4.20mm; Area 2 including AOP – posterior part of anterior olfactory nucleus, AOM – medial part of anterior olfactory nucleus, and AOV – ventral part of anterior olfactory nucleus at Interaural 12.70mm and Bregma 2.70mm; Area 3 including Cg1 – area 1 of cingulate cortex and Cg2 – area 2 of cingulate cortex at Interaural 11.20mm and Bregma 1.70mm; Area 4 including S1J – jaw region of primary somatosensory cortex, GI – granular insular cortex, and AID – dorsal part of agranular insular cortex at Interaural 10.6mm and Bregma 1.00mm; Area 5 including RsGb – retrosplenial granular b cortex and RSA – retrosplenial agranular cortex at Interaural 9.70mm and Bregma 0.20mm; Area 6 including M1 – primary motor cortex and M2 – secondary motor cortex at Interaural 8.70mm and Bregma 0.80mm; Area 7 including S1Tr – trunk region of primary somatosensory cortex and S1DZ –dysgranular region of primary somatosensory cortex at Interaural 8.08mm and Bregma -1.80mm; Area 8 including CeC – capsular part of central amygdaloid nucleus, CeL – lateral division of central amygdaloid nucleus, and CeM – medial division of central amygdaloid nucleus at Interaural 6.88mm and Bregma 3.30mm; Area 9 including CA1 – field CA1 of hippocampus, CA2 – field CA2 of hippocampus, and CA3 – field CA3 of hippocampus at Interaural 5.70mm and Bregma 4.30mm.
Cell counting was performed using Image J software (ImageJ, RRID:SCR_003070) with the blue filter to count the sections stained with H&E, and the green filter to count those stained with anti-GFAP.
The cell counting was performed using both stains. In order to obtain the non-GFAP count, we subtracted the anti-GFAP cell count from the absolute count observed in the H&E slides at the same analyzed area. All the collected data were analyzed using Student’s t-test, performed for each group according to experimental group, subject’s sex, brain area, and staining of the slide. Statistical analyses were carried out based on a population of 10 subjects per experimental and sex groups. We used an alpha level of .05 for all statistical tests.
Results
Regarding GFAP marked cell count in males shown in Figure 1, differences were found in Areas 2, 4, 6, 8 but only reaching strong correlation in the motor cortex located in Area 6 t (1,83) = 3,92, p= <0,01, and amygdala cortex located in Area 9 t (1,83) = 2,18, p= 0,03.
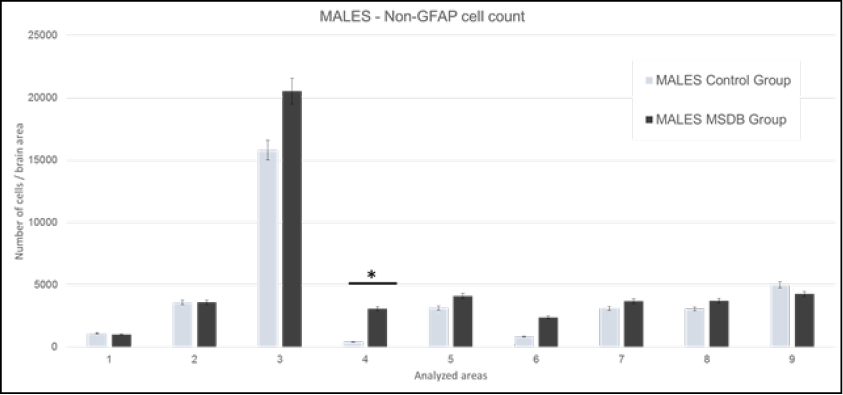
Figure 1. Males: Mean of GFAP marked cells in analyzed areas. Data plotted with 2 SD error bar. 1: M1 - primary motor cortex, LO - lateral orbital cortex, and VO - ventral orbital cortex. 2: AOP - posterior part of anterior olfactory nucleus, AOM - medial part of anterior olfactory nucleus, and AOV - ventral part of anterior olfactory nucleus. 3: Cg1 - area 1 of cingulate cortex, and Cg2 - area 2 of cingulate cortex. 4: S1J - jaw region of primary somatosensory cortex, GI - granular insular cortex, and AID - dorsal part of agranular insular cortex. 5: RsGb - retrosplenial granular b cortex, and RSA - retrosplenial agranular cortex. 6: M1 - primary motor cortex, and M2 - secondary motor cortex. 7: S1Tr – trunk region of primary somatosensory cortex and S1DZ –dysgranular region of primary somatosensory cortex. 8: CeC – capsular part of central amygdaloid nucleus, CeL – lateral division of central amygdaloid nucleus, and CeM – medial division of central amygdaloid nucleus. 9: CA1 - field CA1 of hippocampus, CA2 - field CA2 of hippocampus, and CA3 - field CA3 of hippocampus. Source: authors.
GFAP marked cells count in female subjects is shown in Figure 2, strong statistically correlation was found in: Area 1 t(1,83)= 5,40, p= <0,01; Area 2 t(1,83)= 3,91, p= <0,01; Area 3 t(1,83)= 2,21, p= 0,03; Area 5 t(1,83)= 6,01, p= <0,01; Area 6 t(1,83)= 4,21, p= <0,01; and Area 9 t(1,83)= 3,64, p= <0,01. These areas include orbital, olfactory, cingulate, retrosplenial, motor, and hippocampus cortices, respectively.
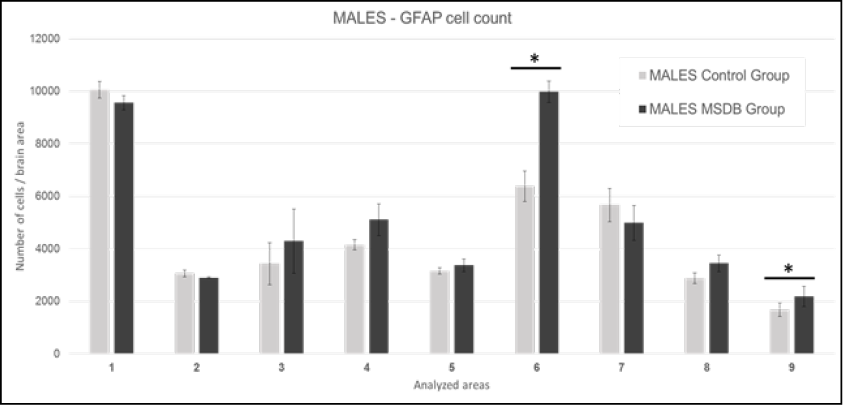
Figure 2. Females: Mean of GFAP marked cells in analyzed areas. Data is presented with 2 SD error bar. 1: M1 - primary motor cortex, LO - lateral orbital cortex, and VO - ventral orbital cortex. 2: AOP - posterior part of anterior olfactory nucleus, AOM - medial part of anterior olfactory nucleus, and AOV - ventral part of anterior olfactory nucleus. 3: Cg1 - area 1 of cingulate cortex, and Cg2 - area 2 of cingulate cortex. 4: S1J - jaw region of primary somatosensory cortex, GI - granular insular cortex, and AID - dorsal part of agranular insular cortex. 5: RsGb - retrosplenial granular b cortex, and RSA - retrosplenial agranular cortex. 6: M1 - primary motor cortex, and M2 - secondary motor cortex. 7: S1Tr – trunk region of primary somatosensory cortex and S1DZ –dysgranular region of primary somatosensory cortex. 8: CeC – capsular part of central amygdaloid nucleus, CeL – lateral division of central amygdaloid nucleus, and CeM – medial division of central amygdaloid nucleus. 9: CA1 - field CA1 of hippocampus, CA2 - field CA2 of hippocampus, and CA3 - field CA3 of hippocampus. Source: authors.
The cell count of non-GFAP cells was calculated as previously detailed, data of male subjects are shown in Figure 3. Differences were found in Areas 3, 4, 5, 6, 9, but taking into consideration the previous GFAP and H&E cell counts with their respective statistically significances, we reached strong correlation only in Area 4. This area includes the insular cortex.
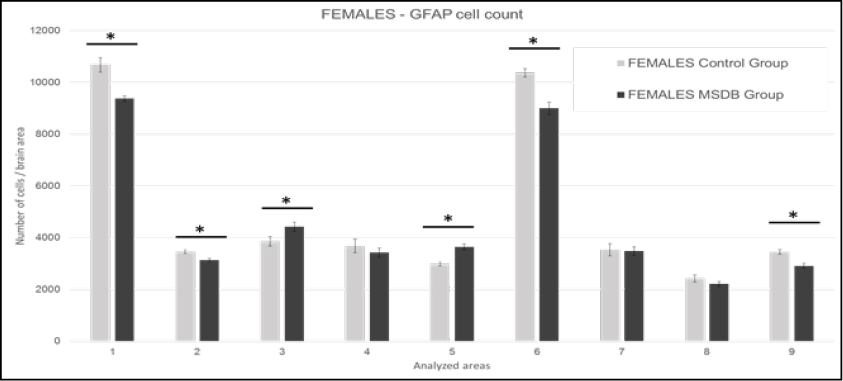
Figure 3. Males: Mean of non-GFAP cells in analyzed areas. Data is presented with 0.05 error bar. 1: M1 - primary motor cortex, LO - lateral orbital cortex, and VO - ventral orbital cortex. 2: AOP - posterior part of anterior olfactory nucleus, AOM - medial part of anterior olfactory nucleus, and AOV - ventral part of anterior olfactory nucleus. 3: Cg1 - area 1 of cingulate cortex, and Cg2 - area 2 of cingulate cortex. 4: S1J - jaw region of primary somatosensory cortex, GI - granular insular cortex, and AID - dorsal part of agranular insular cortex. 5: RsGb - retrosplenial granular b cortex, and RSA - retrosplenial agranular cortex. 6: M1 - primary motor cortex, and M2 - secondary motor cortex. 7: S1Tr – trunk region of primary somatosensory cortex and S1DZ –dysgranular region of primary somatosensory cortex. 8: CeC – capsular part of central amygdaloid nucleus, CeL – lateral division of central amygdaloid nucleus, and CeM – medial division of central amygdaloid nucleus. 9: CA1 - field CA1 of hippocampus, CA2 - field CA2 of hippocampus, and CA3 - field CA3 of hippocampus. Source: authors.
Regarding female subjects, data for non-GFAP cells is shown in Figure 4. Differences were found in Areas 1, 2, 4, 5, 7, 9, but taking into consideration the statistics values for significance in previous anti-GFAP and H&E cell counts, strong correlation was only found in Areas 1, 2, and 9. These areas include the orbital, olfactory, and hippocampus cortices, respectively.
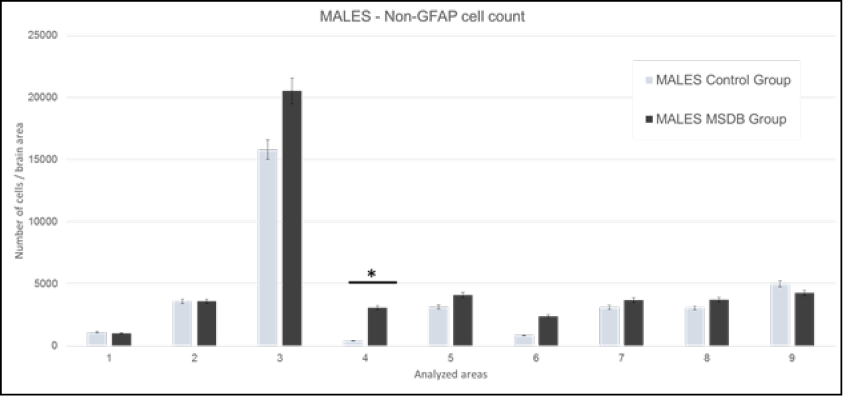
Figure 4. Females: Mean of non-GFAP cells in analyzed areas. Data is presented with 0.05 error bar. 1: M1 - primary motor cortex, LO - lateral orbital cortex, and VO - ventral orbital cortex. 2: AOP - posterior part of anterior olfactory nucleus, AOM - medial part of anterior olfactory nucleus, and AOV - ventral part of anterior olfactory nucleus. 3: Cg1 - area 1 of cingulate cortex, and Cg2 - area 2 of cingulate cortex. 4: S1J - jaw region of primary somatosensory cortex, GI - granular insular cortex, and AID - dorsal part of agranular insular cortex. 5: RsGb - retrosplenial granular b cortex, and RSA - retrosplenial agranular cortex. 6: M1 - primary motor cortex, and M2 - secondary motor cortex. 7: S1Tr – trunk region of primary somatosensory cortex and S1DZ –dysgranular region of primary somatosensory cortex. 8: CeC – capsular part of central amygdaloid nucleus, CeL – lateral division of central amygdaloid nucleus, and CeM – medial division of central amygdaloid nucleus. 9: CA1 - field CA1 of hippocampus, CA2 - field CA2 of hippocampus, and CA3 - field CA3 of hippocampus. Source: authors.
Discussion
The neurobiological consequences of maternal separation during breastfeeding have been approached using animal models. In fact, alterations of the axis that regulates the response to stress have been reported using MSDB model in rats [8]. Our research group has contributed studies regarding behavioral changes of the separated subjects and differences associated with subject’s sex [18, 20].
In this study, statistical significance was obtained at various cortices for both sexes. In GFAP-marked cells count of male subjects, changes were noticed at the motor and hippocampus cortices, Areas 6 and 9, respectively; meanwhile, in non-GFAP cells count, changes were found at the insular and motor cortices, Areas 4 and 6, respectively. However, in GFAP-marked cells count of female subjects, variation was observed at the orbital, olfactory, cingulate, retrosplenial, motor, and hippocampus cortices, Areas 1, 2, 3, 5, 6, 9, respectively; while in non-GFAP cells count, differences were detected at the orbital, olfactory, and hippocampus cortices, Areas 1, 2, and 9, respectively.
The most evident differences were found in counts of cells marked with anti-GFAP in both sexes along the primary motor and hippocampus cortices. The hippocampus is recognized as one of the principal areas in control of the response to stress, learning, and memory [27]. Early studies with other maternal separation protocols, indicated the presence of changes in the architecture of the hippocampus, specifically in CA1, CA3 and the dentate gyrus [12, 28, 29] as a consequence of chronic stress, as well as reduction in the abilities of learning and memory during adult life [27]; moreover, a predisposition to stress-related disorders characterized by disruption in glucocorticoid secretion [30]. The results at the hippocampal area, indicate a higher cell count for marked and non-marked GFAP cells in male subjects, on the other hand, female subjects showed less cell count for marked and non-marked cells; this discrepancy between sex groups has been largely found in previous studies from our research group and other groups. Previous articles from our research group have explained other details addressing the changes in the hippocampus cell count [18, 20].
Indeed, no other previous studies have carried out a specific cell count for GFAP-marked cells along the primary motor cortex Discrepancy was observed regarding the direction of the changes between sex groups. For male subjects, there was an increase in GFAP-marked cells in primary motor cortices, M1 and M2; in contrast, for female subjects, there was a GFAP-marked cells count decrease in these same areas. Some articles have detailed the morphometrical changes in the gross anatomical thickness of these cortices [31], but using a protocol of maternal deprivation that is not fully related to the MSDB protocol used in this study. Many authors have shown the association between changes in the cell counting and other alterations regarding cellular structure and metabolism [32], the regulation of nervous impulses [27], and behavioral changes related with anxiety [17]. Recent studies described specific changes in interneuron maturation triggered by MSDB protocol, also contributing to decrease the total cell count in analyzed cortices [33], leading to the possibility of a sequence of events started by MSDB and ending in cortical dysfunction of this areas.
The findings of this study are probably associated with changes in the HPA axis response related to alterations in corticosterone levels, as well as motor responses to anxiety [6, 8, 17, 34]; subsequently, these changes could also involve modifications in the mechanisms of response to anxiety. In addition, several articles have related connectivity between the hippocampus and the amygdala, including their association with anxiety disorders [35]. Previous findings by other authors [8, 19, 36], suggested that changes in glial cells and neurons in areas connected with the amygdala are a possible adaptive mechanism to stress with a later increase of outputs towards the amygdala nuclei. It is important to note that these increases in GFAP-marked cell count were not observed in female subjects, indicating the presence of other factors affecting the GFAP-positive cells population, leading to a more complex needing for understanding the role of those cells in this MSDB protocol scenario, according to the subject’s sex group.
Moreover, changes in immunoreactivity to GFAP imply modifications in the neuroglia that can be linked directly to the astrocytes and its maturation probably affected by the MSDB protocol; on the other hand, as part of the limitations of this study, this changes in GFAP-marked cell may be partially caused by brain repair activity due to the fundamental role of glial cells to keep the ionic equilibrium, nutrition, and stability of synaptic communication in the brain, as part of the process known as reactive gliosis [37].
The findings of this study should be reinforced by other studies with larger sample size, and specific staining to differentiate the cell types summarized here as non-GFAP, allowing also a greater number of sections to be analyzed. It is important to conduct further studies that correlate the motor cortex alterations found in this study and the motor response to stress, carrying out behavioral studies that aim to unveil the possible role of these cell count changes in the alteration of the motor neural circuits and subsequently transforming the motor response to stress, altered motor response largely seen in rodent subjects exposed to MSDB protocols.
Conflict of interests: none declared by the authors.
Funding sources: This research was financially supported by the Convocatoria Programa Nacional de Semillero De Investigación, Creación E Innovación of the Universidad Nacional de Colombia. –HERMES- 20132015 Código: 20529.
Cited literature
1. Heim C, Nemeroff CB. The role of childhood trauma in the neurobiology of mood and anxiety disorders: preclinical and clinical studies. Biol Psychiatry 2001; 49(12):1023-39. DOI: 10.1016/ s0006-3223(01)01157-x.
2. Humphreys KL, Zeanah CH. Deviations from the expectable environment in early childhood and emerging psychopathology. Neuropsychophar- macology 2015; 40(1):154-70. DOI: 10.1038/ npp.2014.165.
3. Mok PLH, Astrup A, Carr MJ, Antonsen S, Webb RT, Pedersen CB. Experience of Child-Parent Separation and Later Risk of Violent Criminality. Am J Prev Med 2018; 55(2):178-86. DOI: 10.1016/j. amepre.2018.04.008.
4. Saleh A, Potter GG, McQuoid DR, Boyd B, Turner R, MacFall JR, et al. Effects of early life stress on depression, cognitive performance and brain morphology. Psychol Med 2017; 47(1):171-81. DOI:
10.1017/s0033291716002403.
5. Dalsant A, Truzzi A, Setoh P, Esposito G. Maternal bonding in childhood moderates autonomic responses to distress stimuli in adult males. Behav Brain Res 2015; 292:428-31. DOI: 10.1016/j. bbr.2015.06.026.
6. Lippmann M, Bress A, Nemeroff CB, Plotsky PM, Monteggia LM. Long-term behavioural and mole- cular alterations associated with maternal sepa- ration in rats. Eur J Neurosci 2007; 25(10):3091-8. DOI: 10.1111/j.1460-9568.2007.05522.x.
7. Liu J, Leung P, Yang A. Breastfeeding and active bonding protects against children’s internalizing behavior problems. Nutrients 2013; 6(1):76-89. DOI: 10.3390/nu6010076.
8. Pesonen AK, Raikkonen K, Feldt K, Heinonen K, Osmond C, Phillips DI, et al. Childhood separation experience predicts HPA axis hormonal respon- ses in late adulthood: a natural experiment of World War II. Psychoneuroendocrinology 2010;
35(5):758-67. DOI: 10.1016/j.psyneuen.2009.10.017.
9. Rege M, Solli IF. The impact of paternity leave on fathers’ future earnings. Demography 2013;
50(6):2255-77. DOI: 10.1007/s13524-013-0233-1.
10. Carlyle BC, Duque A, Kitchen RR, Bordner KA, Co- man D, Doolittle E, et al. Maternal separation with early weaning: a rodent model providing novel insights into neglect associated developmental deficits. Dev Psychopathol 2012; 24(4):1401-16. DOI: 10.1017/s095457941200079x.
11. Champagne FA, Curley JP. How social expe- riences influence the brain. Current opinion in neurobiology 2005; 15(6):704-9. DOI: 10.1016/j. conb.2005.10.001.
12. Lupien SJ, Parent S, Evans AC, Tremblay RE, Zelazo PD, Corbo V, et al. Larger amygdala but no change in hippocampal volume in 10-year-old children exposed to maternal depressive symptomatology since birth. Proceedings of the National Academy of Sciences of the United States of America 2011;
108(34):14324-9. DOI: 10.1073/pnas.1105371108.
13. Nishi M, Horii-Hayashi N, Sasagawa T. Effects of early life adverse experiences on the brain: implications from maternal separation models in rodents. Front Neurosci 2014; 8:166. DOI: 10.3389/ fnins.2014.00166.
14. Jankord R, Herman JP. Limbic regulation of hy- pothalamo-pituitary-adrenocortical function du- ring acute and chronic stress. Ann N Y Acad Sci
2008; 1148:64-73. DOI: 10.1196/annals.1410.012.
15. Plotsky PM, Meaney MJ. Early, postnatal experien- ce alters hypothalamic corticotropin-releasing factor (CRF) mRNA, median eminence CRF content and stress-induced release in adult rats. Brain Res Mol Brain Res 1993; 18(3):195-200. DOI:
10.1016/0169-328x(93)90189-v.
16. Slotten HA, Kalinichev M, Hagan JJ, Marsden CA, Fone KC. Long-lasting changes in behavioural and neuroendocrine indices in the rat following neonatal maternal separation: gender-dependent effects. Brain Res 2006; 1097(1):123-32. DOI:
10.1016/j.brainres.2006.04.066.
17. Caldji C, Francis D, Sharma S, Plotsky PM, Meaney MJ. The effects of early rearing environment on the development of GABAA and central benzodiazepine receptor levels and novelty- induced fearfulness in the rat. Neuropsychophar- macology 2000; 22(3):219-29. DOI: 10.1016/s0893133x(99)00110-4.
18. Leon Rodriguez DA, Duenas Z. Maternal Sepa- ration during Breastfeeding Induces Gender- Dependent Changes in Anxiety and the GABA-A Receptor Alpha-Subunit in Adult Wistar Rats. PLoS One 2013; 8(6):e68010. DOI: 10.1371/journal. pone.0068010.
19. Caldji C, Tannenbaum B, Sharma S, Francis D, Plotsky PM, Meaney MJ. Maternal care during infancy regulates the development of neural systems mediating the expression of fearfulness in the rat. Proceedings of the National Academy of Sciences of the United States of America. 1998;
95(9):5335-40. DOI: 10.1073/pnas.95.9.5335.
20. Bautista E, Dueñas Z. Maternal separation during breastfeeding induces changes in the number of cells immunolabeled to GFAP. J Psychology& Neuroscience 2012; 5:207-13. DOI: 10.3922/j. psns.2012.2.11.
21. Hsu FC, Zhang GJ, Raol YS, Valentino RJ, Coul- ter DA, Brooks-Kayal AR. Repeated neonatal handling with maternal separation permanently alters hippocampal GABAA receptors and be- havioral stress responses. Proceedings of the National Academy of Sciences of the United States of America 2003; 100(21):12213-8. DOI: 10.1073/ pnas.2131679100.
22. Baldy C, Fournier S, Boisjoly-Villeneuve S, Tremblay ME, Kinkead R. The influence of sex and neona- tal stress on medullary microglia in rat pups. Exp Physiol 2018; 103(9):1192-9. DOI: 10.1113/ ep087088.
23. Yamawaki Y, Nishida M, Harada K, Akagi H. Data on the effect of maternal separation coupled with social isolation in a forced swim test and gene expression of glial fibrillary acid protein in the prefrontal cortex of rats. Data Brief 2018; 18:496500. DOI: 10.1016/j.dib.2018.03.055.
24. Jeong HS, Lee S, Yoon S, Jung JJ, Cho HB, Kim BN, et al. Morphometric abnormalities of the la- teral ventricles in methamphetamine-dependent subjects. Drug Alcohol Depend 2013; 131(3):222-9. DOI: 10.1016/j.drugalcdep.2013.05.009.
25. Tang X, Holland D, Dale AM, Younes L, Miller MI.The diffeomorphometry of regional shape change rates and its relevance to cognitive deterioration in mild cognitive impairment and Alzheimer’s disease. Hum Brain Mapp 2015; 36(6):2093-117. DOI: 10.1002/hbm.22758.
26. Paxinos G, Watson C. Paxino’s and Watson’s The rat brain in stereotaxic coordinates. 7° ed. Amsterdam; Boston: Elsevier/AP, Academic Press is an imprint of Elsevier; 2014. 1 volume (unpaged) p.
27. McClelland S, Korosi A, Cope J, Ivy A, Baram TZ.Emerging roles of epigenetic mechanisms in the enduring effects of early-life stress and experien- ce on learning and memory. Neurobiol Learn Mem
2011; 96(1):79-88. DOI: 10.1016/j.nlm.2011.02.008.
28. Huot RL, Plotsky PM, Lenox RH, McNamara RK.Neonatal maternal separation reduces hippo- campal mossy fiber density in adult Long Evans rats. Brain Res 2002; 950(1-2):52-63. DOI: 10.1016/ s0006-8993(02)02985-2.
29. Jahanshahi M, Sadeghi Y, Hosseini A, Naghdi N, Marjani A. The effect of spatial learning on the number of astrocytes in the CA3 subfield of the rat hippocampus. Singapore Med J 2008;
49(5):388-91.
30. Renard GM, Rivarola MA, Suarez MM. Gender- dependent effects of early maternal separation and variable chronic stress on vasopressinergic activity and glucocorticoid receptor expression in adult rats. Dev Neurosci 2010; 32(1):71-80. DOI:
10.1159/000280102.
31. Aksic M, Radonjic NV, Aleksic D, Jevtic G, Markovic B, Petronijevic N, et al. Long-term effects of the maternal deprivation on the volume and number of neurons in the rat neocortex and hippocam- pus. Acta Neurobiol Exp (Wars) 2013; 73(3):394-
403. DOI: 10.1155/2014/235238.
32. Drevets WC, Price JL, Furey ML. Brain structural and functional abnormalities in mood disorders: implications for neurocircuitry models of depres- sion. Brain Struct Funct 2008; 213(1-2):93-118. DOI:
10.1007/s00429-008-0189-x.
33. Katahira T, Miyazaki N, Motoyama J. Immediate effects of maternal separation on the deve- lopment of interneurons derived from medial ganglionic eminence in the neonatal mouse hippocampus. Dev Growth Differ 2018; 60(5):278-
90. DOI: 10.1111/dgd.12540.
34. Lundberg S, Martinsson M, Nylander I, Roman E. Altered corticosterone levels and social play behavior after prolonged maternal separation in adolescent male but not female Wistar rats. Horm Behav 2017; 87:137-44. DOI: 10.1016/j. yhbeh.2016.11.016.
35. Etkin A, Schatzberg AF. Common abnormalities and disorder-specific compensation during implicit regulation of emotional processing in generalized anxiety and major depressive disor- ders. Am J Psychiatry 2011; 168(9):968-78. DOI:
10.1176/appi.ajp.2011.10091290.
36. Diehl LA, Pereira Nde S, Laureano DP, Benitz AN, Noschang C, Ferreira AG, et al. Contextual fear conditioning in maternal separated rats: the amygdala as a site for alterations. Neurochem Res 2014; 39(2):384-93. DOI: 10.1007/s11064-013
1230-x.
37. Chang L, Alicata D, Ernst T, Volkow N. Structural and metabolic brain changes in the striatum as- sociated with methamphetamine abuse. Addiction
2007; 102 (Suppl 1):16-32. DOI:10.1111/j.13600443.2006.01782.x.
